Checkmate 9la study design9/7/2023  Patients in this subanalysis were enrolled at 16 centers (14 centers in Japan and 2 in China). Briefly, eligible patients were aged ≥ 18 years with treatment-naive, histologically confirmed stage IV or recurrent NSCLC, an Eastern Cooperative Oncology Group (ECOG) performance status of 0–1, and no sensitizing EGFR or ALK mutations. The study design for CheckMate 9LA (NCT03215706) has been described previously. Here, we report efficacy and safety data for the Asian subpopulation in CheckMate 9LA. It is therefore important to evaluate the clinical activities of therapies in Asian populations to optimize treatment strategies. 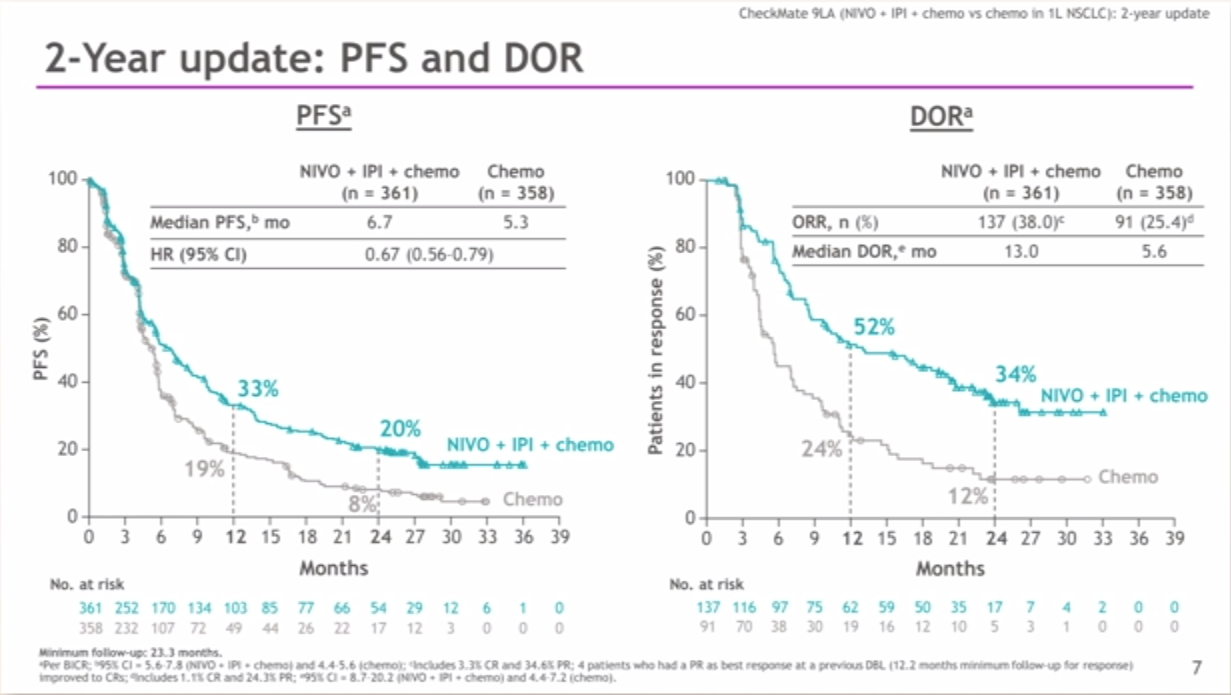
ĭifferences in the treatment outcomes and safety profiles of various therapies have been observed between Asian and non-Asian patients with NSCLC. These data led to the approval of nivolumab plus ipilimumab combined with chemotherapy (2 cycles) in many countries, including the United States, the European Union, and several Asian countries such as Japan, South Korea, Taiwan, and Singapore, as first-line treatment for patients with metastatic or recurrent NSCLC, with no EGFR or ALK genomic tumor aberrations. CheckMate 9LA (NCT03215706), a phase 3, randomized, open-label study in first-line advanced NSCLC, showed significantly improved overall survival (OS), progression-free survival (PFS), and objective response rate (ORR) with nivolumab plus ipilimumab combined with 2 cycles of chemotherapy versus 4 cycles of chemotherapy alone, with a manageable safety profile. 
Addition of a limited course (2 cycles) of platinum-doublet chemotherapy to nivolumab plus ipilimumab was hypothesized to provide rapid initial disease control while potentially building on the durable benefits of nivolumab plus ipilimumab. įirst-line treatment with nivolumab plus ipilimumab provides durable long-term survival benefit for patients with advanced NSCLC, regardless of tumor PD-L1 expression and histology, as observed in CheckMate 227. The immunotherapy agents nivolumab and ipilimumab have distinct but complementary mechanisms of action: nivolumab, a fully human immunoglobulin G4 programmed death (PD)-1 immune checkpoint inhibitor antibody, restores anti-tumor T-cell function and ipilimumab, a fully human immunoglobulin G1 cytotoxic T-lymphocyte antigen-4 immune checkpoint inhibitor antibody, induces T-cell proliferation and de novo anti-tumor T-cell responses, including an increase in memory T cells. Immunotherapy treatments are now approved as monotherapy or in combination with other immunotherapy agents or chemotherapy for the first-line treatment of advanced NSCLC in many countries. ConclusionĬonsistent with results in the all randomized population, nivolumab plus ipilimumab combined with chemotherapy improved efficacy in the Asian subpopulation versus chemotherapy alone and had a manageable safety profile, supporting its use as first-line treatment for advanced NSCLC in Asian patients.Ĭlinical trials of immunotherapy regimens have demonstrated improvements in clinical outcomes over traditional chemotherapy for patients with advanced non-small cell lung cancer (NSCLC). Grade 3–4 treatment-related adverse events were observed in 57% versus 60% of patients, respectively. At a minimum follow-up of 12.7 months, median OS was not reached with nivolumab plus ipilimumab combined with chemotherapy versus 13.3 months with chemotherapy. 
Twenty-eight patients received nivolumab plus ipilimumab combined with chemotherapy and 30 received chemotherapy. Primary endpoint was OS secondary endpoints included progression-free survival (PFS) and objective response rate (ORR). Patients aged ≥ 18 years with treatment-naive, histologically confirmed stage IV or recurrent NSCLC, Eastern Cooperative Oncology Group performance status 0–1 and no sensitizing EGFR/ALK mutations were randomized 1:1 to nivolumab plus ipilimumab (1 mg/kg Q6W) combined with chemotherapy (Q3W for 2 cycles), or chemotherapy alone (Q3W for 4 cycles). We present results for the Asian subpopulation enrolled in Japan and China. CheckMate 9LA, a phase 3, randomized, open-label study in first-line advanced non-small cell lung cancer (NSCLC), showed significantly improved overall survival (OS) with nivolumab plus ipilimumab combined with 2 cycles of chemotherapy versus chemotherapy alone (4 cycles). 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
